The products were sold at major retailers and may pose contamination risks, with consumers urged to stop using them and check lot numbers.
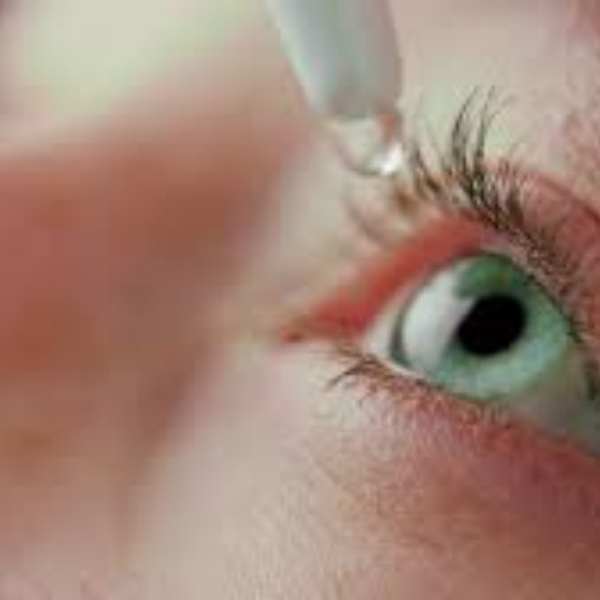
Federal health officials are urging consumers to check their medicine cabinets after more than 3 million bottles of eye drops were recalled nationwide over potential safety concerns.
The U.S. Food and Drug Administration said K.C. Pharmaceuticals, Inc., based in Pomona, California, voluntarily recalled about 3.1 million bottles of various eye drop products due to a “lack of assurance of sterility.”
This means the company cannot confirm the products were manufactured in a way that prevents contamination from bacteria or other microbes.
The FDA classified the recall as Class II, indicating that use of the products could cause temporary or medically reversible health effects.
The affected products include several types of eye drops sold under different labels, including artificial tears, advanced relief, and redness relief formulas. They were distributed nationwide through major retailers and suppliers such as Walgreens, CVS, Kroger, H-E-B, and other chains.
The FDA said most of the recalled products carry expiration dates extending into 2026. While no confirmed contamination has been reported, officials warn that the lack of sterility assurance presents a potential risk.
Health officials advise consumers to stop using the recalled products immediately. Shoppers should check labels and lot numbers, especially those beginning with AC, AR, LT, SU, RG, RL, SY, or AT with 2026 expiration dates.
The recalled eye drop products include:
1,023,096 bottles of Dry Eye Relief Eye Drops (glycerin 0.2%, hypromellose 0.2%, polyethylene glycol 400 1%), 0.5 fl. oz. (15 mL), sold at stores including Rite Aid, H-E-B, Meijer, Harris Teeter, and military exchanges.
589,848 bottles of Artificial Tears Sterile Lubricant Eye Drops (polyvinyl alcohol 0.5%, povidone 0.6%), 0.5 fl. oz. (15 mL), sold at Kroger, Publix, Leader, TopCare, Good Neighbor Pharmacy, and Good Sense.
378,144 bottles of Sterile Eye Drops Original Formula (tetrahydrozoline HCl 0.05%), 0.5 fl. oz. (15 mL), sold at Walgreens, Kroger, CVS, H-E-B, Harris Teeter, Dollar General, Circle K, and Leader.
315,144 bottles of Sterile Eye Drops Redness Lubricant (glycerin 0.25%, naphazoline HCl 0.012%), 0.5 fl. oz. (15 mL), sold at Walgreens, Rite Aid, Leader, Equaline, and through Cintas.
303,216 bottles of Eye Drops Advanced Relief (dextran 70 0.1%, polyethylene glycol 400 1%, tetrahydrozoline HCl 0.05%), 0.5 fl. oz. (15 mL), sold at Walgreens, Kroger, and CVS.
245,184 bottles of Ultra Lubricating Eye Drops (polyethylene glycol 400 0.4%, propylene glycol 0.3%), 0.5 fl. oz. (15 mL), sold at Leader and Harris Teeter.
182,424 bottles of Sterile Eye Drops AC (tetrahydrozoline HCl 0.05%, zinc sulfate 0.25%), 0.5 fl. oz. (15 mL), sold at Walgreens, Meijer, and H-E-B.
74,016 bottles of Sterile Eye Drops Soothing Tears (polyethylene glycol 400 0.4%, propylene glycol 0.3%), 0.5 fl. oz. (15 mL), sold at Walgreens and through Rugby Laboratories.
Anyone who has used these products and experiences symptoms such as eye pain, redness, or vision changes should seek medical care.
The FDA said more details, including full product and lot information, are available in its enforcement report.









Leave a Reply